
The production of chemicals by fermenting various sugars is a well-accepted science. Its use ranges from producing beverage alcohol and fuel-ethanol to making citric acid and xantham gum for food uses. However, the high price of sugar and the relatively low cost of competing petroleum-based fuel have kept the production of chemicals mainly confined to producing ethanol from corn sugar – until now.
The Company has developed significant proprietary improvements to a well-known conversion technology known as Concentrated Acid Hydrolysis such that the process is ready for commercial implementation. The Technology is unique in that, for the first time, it enables widely available cellulosic materials, or more commonly, biomass, to be converted into sugar in an economically viable manner, thereby providing an inexpensive raw material for fermentation or chemical conversion into any of a hundred different specialty and/or commodity chemicals.
Proven Process Stability + Economic Viability


It has been known for over 100 years that acids act as catalyst to convert (“hydrolyze”) cellulose and hemicellulose into simple sugars (hexose and pentose, or “C6 and C5” sugars). The Germans and Russians used this simple procedure in the early part of this century to produce alcohol fuels and chemicals from wood in order to supply their war efforts. However they all were not economically competitive with low cost petroleum products because of poor yields, high wastage, and the large volume of unmarketable by-products.
In 1989 Arkenol, began research and determined that the Concentrated Acid Hydrolysis process could be made economically viable through the use of new technology, modern control methods, and newer materials of construction. As a result, Arkenol were able to solve the problems with the following proprietary improvements that now make the process economically viable:
![]()
efficient acid recovery and reconcentration
![]()
high sugar concentration at high purity
![]()
the ability to ferment C6 and C5 sugars efficiently with conventional microbes
![]()
the ability to handle silica in biomass feedstocks
![]()
all by-products are usable and marketable
Arkenol’s improvements to the technology are designed to lower production costs, improve yield and make the process more viable.
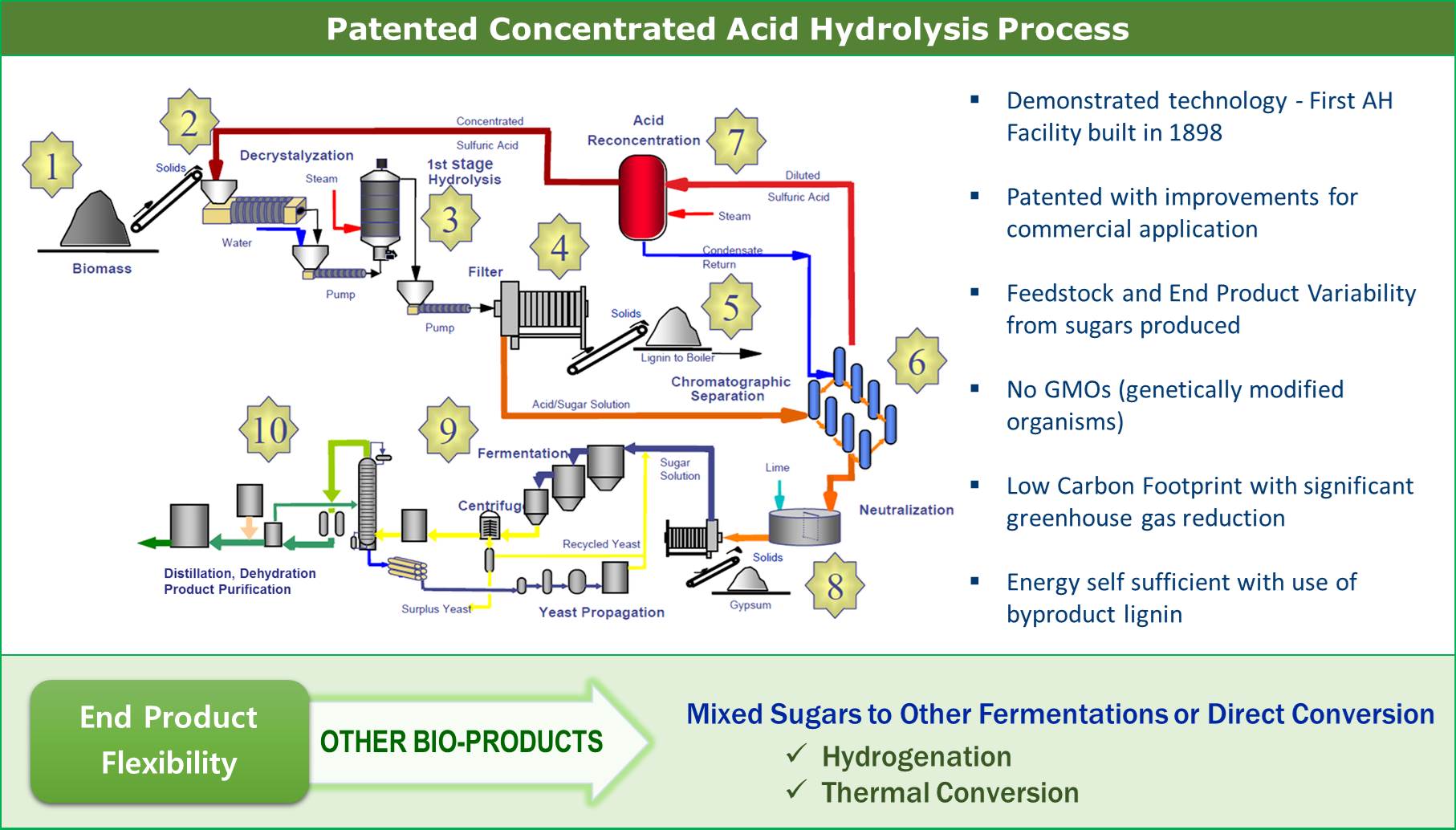
The Process
Arkenol’s proprietary concentrated acid hydrolysis technology converts lignocellulosic materials to value-added industrial and consumer end-products in fuels and chemicals categories. Stage one processes the primary feedstock into fermentable sugars of both five and six carbon atoms, while the second stage of the process takes these fermentable sugars and converts them to the desired fuel or chemical end-products. Likewise, Arkenol’s fermentation systems are based on fermenting both five- and six carbon sugars to value-added end-products. This immensely adds to the overall economic productivity of the process. Conventional fermentation schemes are limited to processing only six-carbon sugar into ethanol, butanol or citric acid.
An integrated, full-scale commercial process plant consists of six basic unit operations:
- – Feedstock preparation
- – Decrystallization/Hydrolysis Reaction Vessel
- – Solids/Liquid Filtration
- – Separation of the acid and sugars
- – Fermentation of the sugars
- – Product purification
Simply put, the process separates the biomass into its main constituents: cellulose and hemicellulose (the main building blocks of plant life) and lignin (the “glue” that holds the building blocks together), then converts the cellulose and hemicellulose to sugars, ferments them and purifies the fermentation liquids into products. These unit operations require a series of material and energy inputs to produce the primary products of fermentation and the resultant by-products.
The advantages of the concentrated acid process are that the reaction is fast and is carried out at lower temperatures and pressures than those using dilute acid. These advantages result in less unwanted degradation products.





 COPYRIGHT © 2016 ARKENOL & AKR. ALL RIGHTS RESERVED.
COPYRIGHT © 2016 ARKENOL & AKR. ALL RIGHTS RESERVED.